About
Improving the care pathway for patients with respiratory failure
Biosency aims to improve the management of patients with cardio-respiratory insufficiency by offering a disruptive digital solution, integrating clinical data analysis, to optimise monitoring and facilitate coordination between health professionals.
Our missions:
- Develop the best remote monitoring solutions
- Develop a solution for predicting COPD exacerbations
- Host and distribute integrated services to exploit the full potential of real-life data for clinical, research and statistical purposes
We are continuing our R&D work to complete our offer and promote prevention through predictive diagnosis or the integration of new measures.

Remote monitoring
Oxygen titration aid
Carrying out regular check-ups
Monitoring of patients at risk

Clinical studies
Real-time and automatic data collection

Predictive diagnosis
Prediction of COPD exacerbations
Founders


Historical

Society creation
July 2017Biosency was created in July 2017 by its 2 founders: Marie Pirotais, Yann Le Guillou. The latter were surrounded by a scientific council of 6 expert pulmonologists.

Winner of the i-Lab trophy
July 2018Biosency is the winner of the 20th i-Lab competition. I-lab is the national competition to support the creation of innovative technological companies, organized by the Ministry of Higher Education, Research and Innovation. It rewards the best French deeptech projects.

Compliance of Bora band®
March 2019Bora band® complies with the standards: ISO 80601-2-61: Performance validated by a clinical study ISO 80601-2-56 clause 201.101.2: Accuracy of skin temperature validated by a laboratory IEC 60601-1, IEC 60601-1- 2 and IEC 60601-1-11: Electrical safety and EMC testing ETSI EN ETSI EN 300 328 V2.1.1 and EN 62479: EMC standard for radio equipment and services IEC 60601-1-1: Validation of home use IEC 60529-1 – IP64: No dust penetration, water resistant SO 10993-5, and 10993-11: Cytotoxicity, sensitization and irritation to assess permanent contact with silicone, polycarbonate, carbon black and stainless steel with intact skin. ISO 60601-1-6: General safety requirements – Collateral standard: Ease of use

Obtaining CE DM marking
June 2019Obtaining certification is a key step in Biosency's development. The CE DM certificate, issued by the notified body BSI, certifies the entire remote monitoring service.
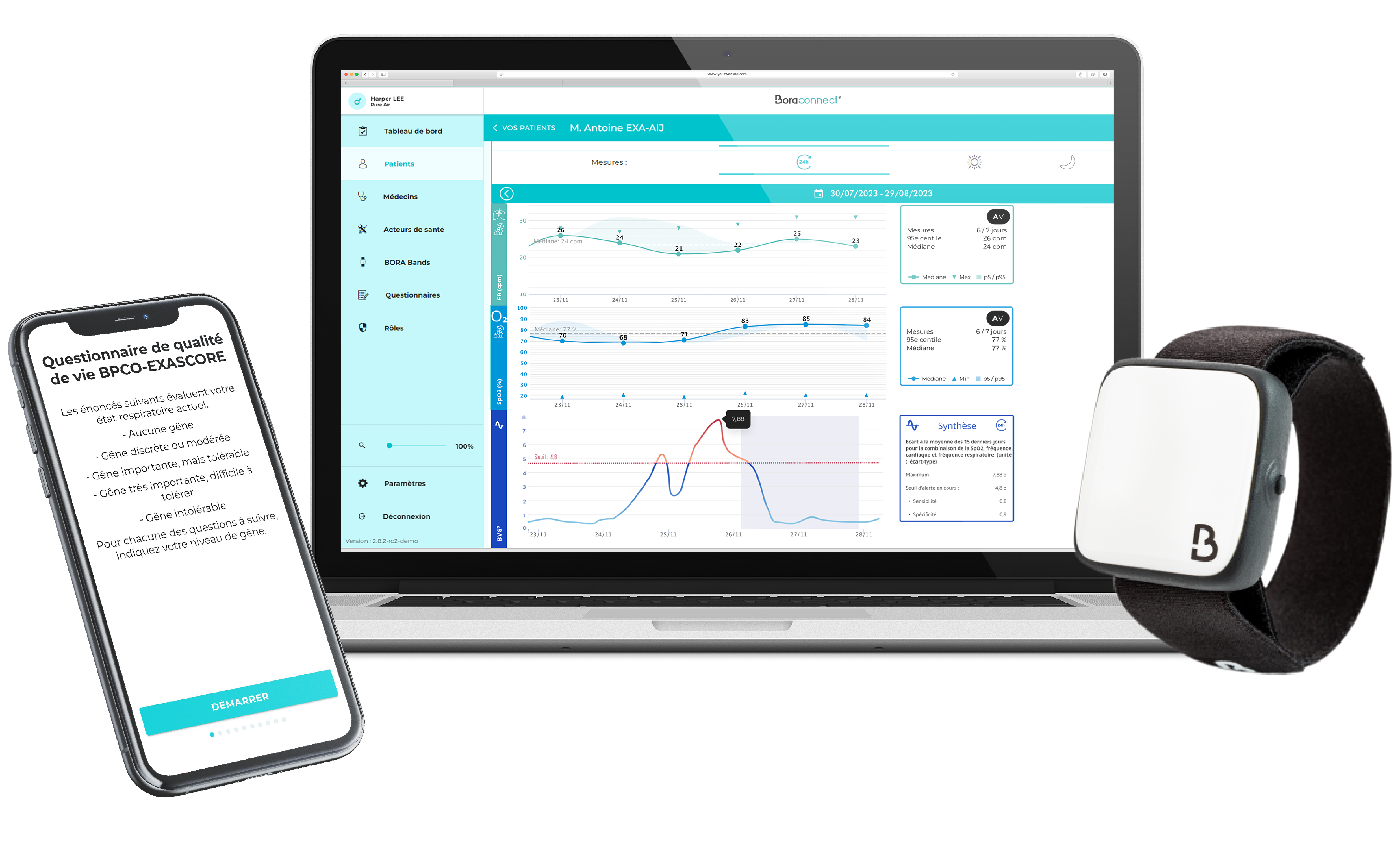
Commercial launch of Bora connect® and Bora band®
2020This step culminates several years of research and development and allows patients and healthcare professionals to benefit from a complete medical remote monitoring solution.

Winner of the i-Nov trophy
February 2020Biosency is the winner of the “Innovation Competition – i-Nov” call for projects in the “deeptech” category. i-nov is a support system financed by the Future Investment Program (PIA) which aims to select innovation projects with particularly strong potential for the French economy.

Inclusion of the first patient in the e-meuse study
September 2021Biosency, Territoire Innovation winner, is participating in the first large-scale remote monitoring experiment for patients suffering from sleep apnea or COPD. For 3 and a half years, 400 patients will be monitored remotely in order to assess the benefit of treatment integrating connected objects in these chronic respiratory diseases.

Strategic partnership with ResMed
October 2022A new major step in our development has been reached with the signing of a strategic partnership with ResMed, world leader in innovative solutions for the treatment of respiratory disorders. ResMed will thus distribute, in France, the Maghreb and certain European countries, our Bora care® remote monitoring solution, to healthcare professionals who care for patients suffering from respiratory failure.
Early detection of exacerbations
May 2023During the prestigious international conference of the American Thoracic Society (ATS 2023) held in Washington, DC, Yann Le Guillou, co-founder of Biosency, revealed the results of his latest clinical study. These highlight the ability of our BVS3 technology to predict acute exacerbations of COPD (chronic obstructive pulmonary disease) on average 3 days before hospitalization in 86% cases, with only 9% false positives.

Obtaining MDR (Medical Device Regulation) certification
June 2023This certification is proof of our dedication to meeting the strict standards and regulations set out under the European Medical Device Regulation and reinforces our commitment to ensuring the highest level of quality, safety and performance for our medical device, for the benefit of patients and healthcare professionals.

Definitive ANS certification
December 2023Biosency has reached a major milestone by obtaining final certification in the interoperability and security framework for digital medical devices for its remote monitoring offering dedicated to patients with respiratory failure.
This achievement represents not only the recognition of our commitment to excellence and quality, data protection but also the validation of the effectiveness of our innovative solution in the field of health.
